CISTAR Technical Highlights
The following highlights summarize significant discoveries in catalysis, separations, and process systems engineering made by CISTAR's researchers.
ALTERNATIVE LIGHT OLEFIN PRODUCTION THROUGH METHANE DILUTED CRACKING, TURBOQUENCHING, AND ELECTRIC CRACKING
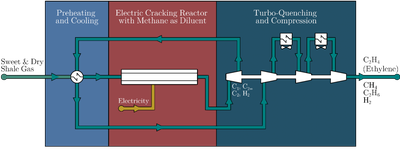 Figure: CISTAR's innovative process for the production of light olefins.
Figure: CISTAR's innovative process for the production of light olefins.
Outcome/accomplishment: CISTAR researchers have introduced an innovative process for the production of light olefins integrating three new concepts: Methane Diluted Cracking, Turboquenching, and Electric Cracking, as illustrated in the Figure.
Impact/benefits: The proposed design offers numerous advantages over the best-known conventional method. By substantially cutting the number of unit operations and streamlining the process, it reduces complexity and enhances efficiency. Additionally, the process requires only one service stream in the form of electricity, eliminating the need for water during quenching, and repeated cryogenic separations at the front-end and back-end of the process. These improvements result in significant reductions in energy consumption, cost, and CO2 emissions. Furthermore, this process is more amenable to modularization and for distributed manufacturing.
Explanation/Background: Olefins are some of the most important chemicals that are used for the manufacturing of a wide array of products with applications spanning a wide range of fields, including medicine, electronics, food contact materials, photovoltaic panels, textiles, and many more. As a result, the efficient production of olefins is of paramount importance for modern society.
For the past century, the production of light olefins has relied on conventional methods such as flame-heated tubular crackers, water as diluent for the cracking process, indirect quenching with water or other fluids, compression with water knockouts, gas drying, and repeated cryogenic separations at the front-end and back-end of the process. However, the new proposed process offers significant improvements and advantages over the conventional methods.
NON-THERMAL PLASMA ACTIVATION AND COUPLING OF LIGHT ALKANES
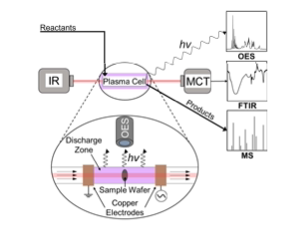 Figure: A dielectric barrier discharge (DBD) plasma cell capable of interfacing with Fourier transform infrared spectroscopy, optical emission spectroscopy, and mass spectrometry to simultaneously characterize the surface, the plasma phase, and the gas phase. Clarke and Hicks, ACS Eng. Au 2022, 2, 6, 535–546
Figure: A dielectric barrier discharge (DBD) plasma cell capable of interfacing with Fourier transform infrared spectroscopy, optical emission spectroscopy, and mass spectrometry to simultaneously characterize the surface, the plasma phase, and the gas phase. Clarke and Hicks, ACS Eng. Au 2022, 2, 6, 535–546
Outcome/accomplishment: Experimentally demonstrated feasibility of converting light alkanes to C2+ products (e.g., ethylene, acetylene, propane/propylene, C4+) at low temperatures through use of electrically generated plasma.
Impact/benefits: Ethylene and propylene are currently produced from steam cracking of ethane, propane or higher hydrocarbons at very high temperatures in cracking furnaces. Steam cracking has a large carbon footprint due to the high temperatures (> 750oC) required. Alternative lower temperature pathways, such as electrochemical processes or non-thermal plasmas would allow production of these valuable building block chemicals at with much lower GHG emissions.
Explanation/ background: Non-thermal plasmas (NTPs) are partially ionized gases characterized by a non-equilibrium in temperature hi energy electrons (>10,000 oK) and low energy ions and neutrals (~ambient temperature). The energetic electrons are the primary driver for promoting gas-phase and or surface reactions through collisions with neutral gas-phase molecules to form electronically, vibrationally, and rotationally excited species as well as radicals.
CISTAR researchers have shown that ethane dehydrogenation can occur under NTP stimulation with conversions up to >99 % and ethylene selectivity as high as 28%. Further, the NTP facilitates carbon-carbon coupling reactions generating propane/propylene as well as other larger molecular weight gas phase species. Methane can be co-fed as a reactant for upgrading to more valuable products and to minimize flaring. The role of the catalyst under NTP conditions is the subject of ongoing investigations.
MODIFIED ZEOLITE OLEFIN OLIGOMERIZATION CATALYSTS WITH HIGH LIFETIME AND PRODUCT SELECTIVITY
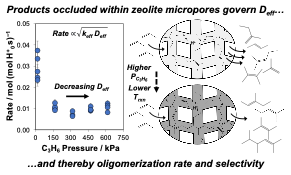 Figure: Hydrocarbon products occlude within MFI zeolite pores during oligomerization catalysis and impose diffusional restrictions that regulate observed reaction rates.
Figure: Hydrocarbon products occlude within MFI zeolite pores during oligomerization catalysis and impose diffusional restrictions that regulate observed reaction rates.
Outcome/accomplishment: Researchers at the Center for Innovative and Strategic Transformation of Alkane Resources (CISTAR) have developed a new acid-zeolite catalysts that catalyze oligomerization reactions of light olefin gases to produce heavier hydrocarbon liquids with superior control over product selectivity and catalyst lifetime.
Impact/benefits: Light olefins, such as ethylene and propylene, can be readily created from ethane and propane that are significant components of natural gas liquids produced in abundance from distributed shale gas resources. Current technical challenges with acid-catalyzed oligomerization of light gases into heavier compounds are the broad molecular weight range of oligomer products that are produced, and the short catalyst lifetimes observed due to the formation of very heavy hydrocarbon products and coke. Catalysts, with improved control over product selectivity and extended lifetime, enable novel reactor and process configurations with lower capital investments that are suitable for distributed manufacturing.
Explanation/ background: Brønsted acid zeolite catalysts provide an attractive materials platform for catalyzing olefin oligomerization reactions, because they can be engineered with different material properties (e.g., active site distributions, micropore size and shape, crystallite length that controls characteristic diffusion parameter) to influence reaction rate, product selectivity and catalyst lifetime. CISTAR researchers have determined that heavier alkene oligomers gradually accumulate within the micropores of medium-pore (ten-membered ring, 10-MR) zeolites during olefin oligomerization catalysis. The composition of these occluded hydrocarbons changes in response to reaction conditions and material properties, imposing intracrystalline diffusion barriers that influence rates of molecular transport. Measured rates and selectivities of olefin oligomerization reflect the combined influences of the kinetic properties of active sites and diffusional constraints imposed by occluded intrapore hydrocarbons, leading to complex dependences of rates and selectivity on alkene pressure and material properties among 10-MR zeolite catalysts (Figure 1). Based on this knowledge, new CISTAR catalysts were developed and patented new catalysts that have improved transport properties leading to higher reaction rates and the formation of liquid products in higher yields with stable time-on-stream compositions.
IMPROVED SHALE GAS ALKANE DIRECT ALKANE AROMATIZATION CATALYST
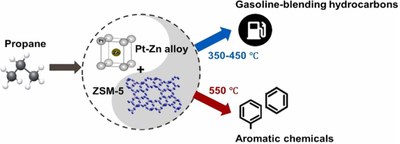 Figure: The temperature dependence on product selectivity for bifunctional conversion of propane to higher molecular weight hydrocarbon leads to different products; Appl. Catal. A, Gen., 643, 118753 (2022): doi: 10.1016/j.apcata.2022.118753.
Figure: The temperature dependence on product selectivity for bifunctional conversion of propane to higher molecular weight hydrocarbon leads to different products; Appl. Catal. A, Gen., 643, 118753 (2022): doi: 10.1016/j.apcata.2022.118753.
Outcome/accomplishment: CISTAR researchers have discovered a novel PtZn/SiO2/MFI zeolite catalyst for direct aromatization of propane and ethane with significantly improved selectivity for aromatics. The new catalyst is the result of fundamental studies of the individual reactions over the catalyst metal and zeolite components, which allowed development of the catalyst with a better balance of metal (dehydrogenation) and MFI olefin conversion rates (aromatic formation).
Impact/benefits: Single ring aromatics, such as benzene, toluene, xylene, and C9+ aromatics, have significant value as both fuel components and chemical feedstocks. Production of aromatics directly from light hydrocarbons like ethane and propane would have significant feed cost advantage vs. today’s production from gasoline range naphtha. The current catalyst for aromatization of propane (Ga on MFI) are limited to yields of 60% and produce significant lower value methane making them un-economic. Preliminary economics by CISTAR suggest that a process based on this catalyst would be economically attractive.
Explanation/ background: Previously, processes to covert propane and butane into aromatics using zinc or gallium-loaded MFI zeolite catalysts have yields near 60%. The BTX (benzene, toluene and xylenes) yields are limited by light gas formation, primarily methane and ethane. Propane aromatization requires two components, the first is a metal (dehydrogenation) component to convert the propane to propene, and the second is a zeolite (MFI) component to oligomerize the propene into higher hydrocarbons which are then dehydro-cyclized into the final aromatic product. CISTAR researchers determined the relative rates and selectivity for the individual reactions for propane conversion on the two catalytic components (metal and zeolite) were determined individually to develop improved catalysts. It was found that in addition catalyzing alkene oligomerization the MFI component also was the primary producer of the light gases (methane and ethane) through mono-molecular acid cracking of the propane. This understanding allowed the CISTAR researchers to lower the amount of the MFI component in the catalyst to significantly reduce the light gas formation. Additionally, an improved PtZn alloy metal component was discovered which has significant selectivity (>99%) and rate advantages (100 times) over the current Ga. The new combined catalyst is a PtZn/SiO2/MFI which give high yields of 80% aromatics with low selectivity to undesirable methane (<5%) at 70% propane conversion. It was also found that the aromatic products can be tuned to either fuels or chemicals by adjusting the reactor temperature.






